| Product: | AICAR | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Catalog Number: | 5391 | ||||||||||||
| CAS Number: | 2627-69-2 | ||||||||||||
| Synonyms: |
5-Aminoimidazole-4-carboxamide 1-β-D-ribofuranoside; N1-(β-D-Ribofuranosyl)-5-aminoimidazole-4-carboxamide; Acadesine |
||||||||||||
| Pricing: |
|
||||||||||||
| Formula: | C9H14N4O5 | ||||||||||||
| Chemical Purity: | >98% | ||||||||||||
| Molecular Weight: | 258.23 | ||||||||||||
| Structure: |
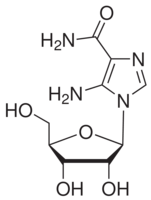
|
||||||||||||
| Appearance: | Powder | ||||||||||||
| Category: | Unlabeled Reference Standards | ||||||||||||
| Stability: | Stable under recommended storage conditions | ||||||||||||
| Storage: |
Keep container tightly closed in a dry and well-ventilated place. Recommended storage temperature: -20°C |
||||||||||||
| Transportation: |
Non-hazardous for transport |
||||||||||||
| Literature References: |
Velásquez, D.A., et al., The Central Sirtuin 1/p53 Pathway Is Essential For The Orexigenic Action Of Ghrelin. Diabetes 60, 1177-85, (2011); Giri, S., et al., 5-Aminoimidazole-4-carboxamide-1-β-4-ribofuranoside inhibits proinflammatory response in glial cells: a possible role of AMP-activated protein kinase. J. Neurosci. 24, 479-487 , (2004); Merck 14,16 |
||||||||||||
| MSDS: | |||||||||||||
| Description: |
AICAR is a cell permeable activator of AMP-activated protein kinase (AMPK), a metabolic master regulator that is activated in times of reduced energy availability (high cellular AMP:ATP ratios) and serves to inhibit anabolic processes. In vivo, pharmacologic activation of AMPK with AICAR mimics exercise and triggers insulin-independent glucose uptake by skeletal muscle |
