| Product: | 2-Fluoroadenine-9-β-D-arabinofuranoside | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Catalog Number: | 32261 | ||||||||
| CAS Number: | 21679-14-1 | ||||||||
| Synonyms: |
9-β-Arabinofuranosyl-2-fluoroadenine; F-ara-A; Fludarabine des-phosphate |
||||||||
| Pricing: |
|
||||||||
| Formula: | C10H12FN5O4 | ||||||||
| Molecular Weight: | 285.23 | ||||||||
| Structure: |
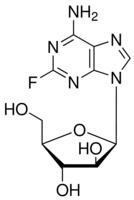
|
||||||||
| Appearance: | White powder | ||||||||
| Category: | Nucleotides and Nucleosides Alphabetic Listing | ||||||||
| Stability: | Stable under recommended storage conditions | ||||||||
| Storage: |
Keep container tightly closed in a dry and well-ventilated place. Recommended storage temperature: 2 - 8°C |
||||||||
| Transportation: |
Non-hazardous for transport |
||||||||
| Literature References: |
Wyczechowska, D., and Fabianowska-Majewska, K., The effects of cladribine and fludarabine on DNA methylation in K562 cells Biochem. Pharmacol. 65, 219-225, (2003); Huang, P., and Plunkett, W. Mol. Pharmacol. 39, 449, (1991); Keating, M.J., et al. Blood 74, 19, (1989); Merck 14,4126 |
||||||||
| MSDS: | |||||||||
| Description: |
Fludarabine (the 5′-phosphate) is a prodrug that is converted to F-ara-A, which enters cells and accumulates primarily as the 5′-triphosphate. F-ara-A interferes with DNA synthesis and repair and induces apoptosis of cancer cells. F-ara-A also strongly inhibits DNA methylation, particularly methylation of cytosine in CpG dinucleotide sequences |
