| Product: | Canrenoic acid potassium salt | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Catalog Number: | 16508 | ||||||||
| CAS Number: | 2181-04-6 | ||||||||
| Synonyms: |
17-Hydroxy-3-oxopregna-4,6-diene-21-carboxylic acid; Potassium canrenoate |
||||||||
| Pricing: |
|
||||||||
| Formula: | C22H30KO4 | ||||||||
| Molecular Weight: | 397.6 | ||||||||
| Structure: |
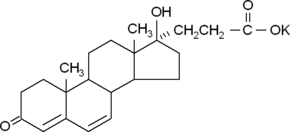
|
||||||||
| Appearance: | Powder | ||||||||
| Category: | Biochemicals and Reagents | ||||||||
| Stability: | Stable under recommended storage conditions. | ||||||||
| Storage: |
Keep container tightly closed in a dry and well ventilated place |
||||||||
| Transportation: |
Non-hazardous for transport |
||||||||
| Literature References: |
Brambilla, G., and Martelli, A., Are some progestins genotoxic liver carcinogens?. Mutat. Res. 512, 155-163, (2002); Young, M., and Funder, J., Mineralocorticoid action and sodium-hydrogen exchange: studies in experimental cardiac fibrosis. Endocrinology 144, 3848-3851, (2003); Martelli, A., et al., Genotoxicity testing of potassium canrenoate in cultured rat and human cells. Mutagenesis 14, 463-472, (1999); Merck 14,1750 |
||||||||
| MSDS: | |||||||||
| Description: |
Competitive aldosterone receptor antagonist. Potassium canrenoate reduces the effects of aldosterone-induced increases in blood pressure and in cardiovascular fibrosis in animals with high sodium intake. It is used clinically for its anti-fibrotic effects. At higher doses it is genotoxic to liver and increases tumor incidence in rodent models. |
