| Product: | Dabigatran etexilate | ||||
|---|---|---|---|---|---|
| Catalog Number: | 18290 | ||||
| CAS Number: | 211915-06-9 | ||||
| Pricing: |
|
||||
| Formula: | C34H41N7O5 | ||||
| Chemical Purity: | >98% | ||||
| Molecular Weight: | 627.73324 | ||||
| Structure: |
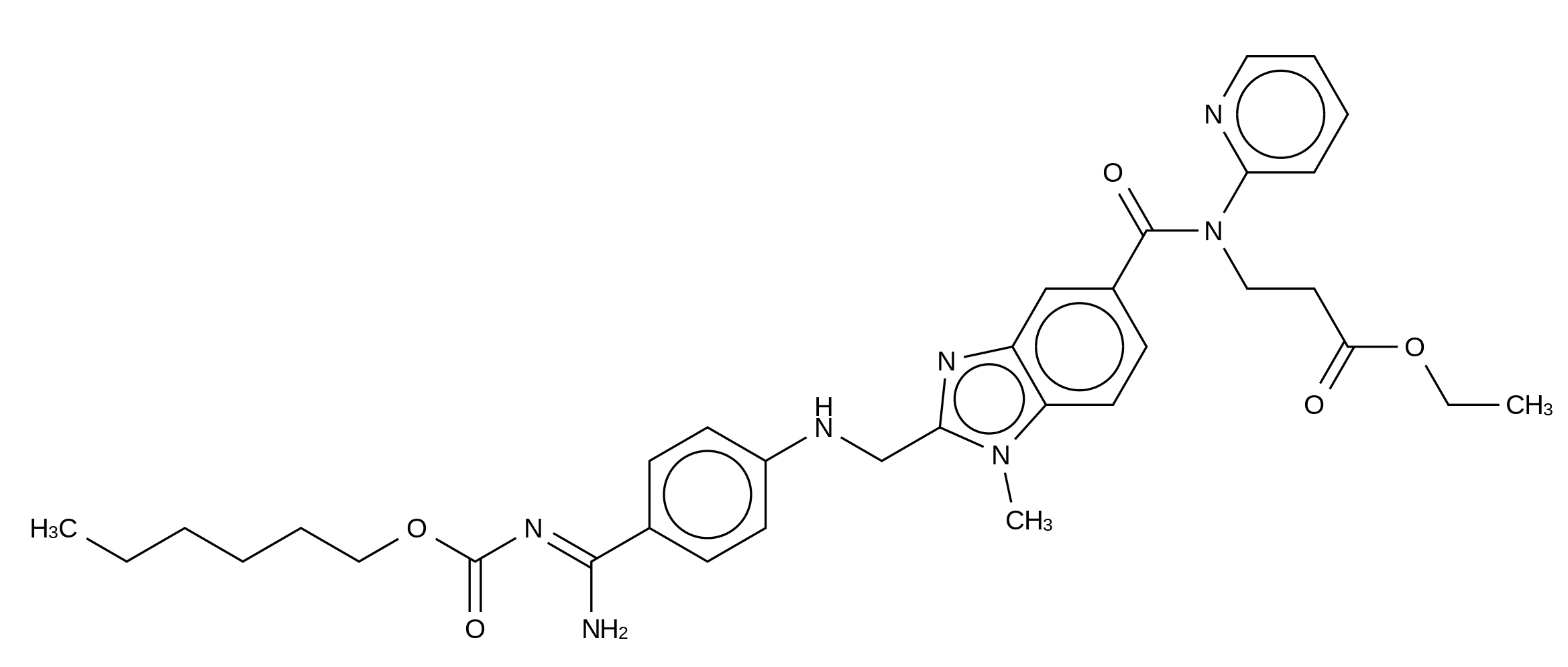
|
||||
| Category: | API Compounds | ||||
| Storage: |
Store at 4° C |
||||
| Literature References: |
Verma, A.K. 2010. Med. J. Aust. 192: 407-412. PMID: 20367591; Walenga, J.M. and Adiguzel, C. 2010. Int. J. Clin. Pract. 64: 956-967. PMID: 20584229; Maegdefessel, L., et al. 2010. Vasc Health Risk Manag. 6: 339-349. PMID: 20531953 |
||||
| MSDS: | |||||
| Description: |
Dabigatran etexilate is the prodrug of the active moiety dabigatran. Dabigatran etexilate has recently been in clinical trials for use as an anticoagulant drug. It has been shown to have slight, but significant drug-drug interactions when used in combination with strong P-glycoprotein inducers or inhibitors such as amiodarone or rifampicin. It has also been associated with lower rates of stroke and systemic embolism than warfarin, a comparable anticoagulant |
