| Product: | Allopurinol | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Catalog Number: | 6298 | ||||||||
| CAS Number: | 315-30-0 | ||||||||
| Synonyms: | 4-Hydroxypyrazolo[3,4-d]pyrimidine; HPP; 1H-Pyrazolo(3,4-d)pyrimidin-4-ol; 4-Hydroxypyrazolo(3,4-d)pyrimidine | ||||||||
| Pricing: |
|
||||||||
| Formula: | C5H4N4O | ||||||||
| Chemical Purity: | 98% | ||||||||
| Molecular Weight: | 136.11 | ||||||||
| Structure: |
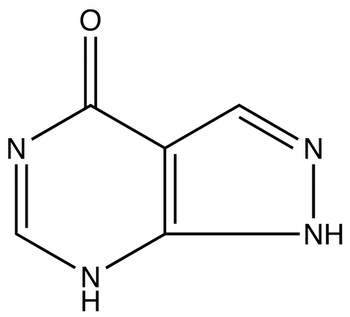
|
||||||||
| Appearance: | Solid | ||||||||
| Category: | Heterocyclic Compounds | ||||||||
| Stability: | Stable under recommended storage conditions | ||||||||
| Storage: |
Keep container tightly closed in a dry and well-ventilated place |
||||||||
| Transportation: |
IATA: Hazard Class: 6.1; UN Number: UN2811; Packing Group: III; Shipping Name: Toxic solid, organic, n.o.s. (Allopurinol) |
||||||||
| Literature References: |
Murrell, G.A, and Rapeport, W.G., Clinical pharmacokinetics of allopurinol Clin. Pharmacokinet. 11, 343-353, (1986); Kelley, W.N. and Beardmore, T.D. Science 169, 388, (1970); Gibbings, S., et al., Xanthine Oxidoreductase Promotes The Inflammatory State Of Mononuclear Phagocytes Through Effects On Chemokine Expression, Peroxisome Proliferator-activated Receptor-{gamma} Sumoylation, And HIF-1{alpha}. J. Thorac. Cardiovasc. Surg. 286, 961-75, (2011); Merck 14,279; FT-IR 2 (3), 3665:C / FT-IR 1 (2), 708:D / IR-Spectra (3), 1286:C / IR-Spectra (2), 1119:B / NMR-Reference 2 (2), 581:C / RegBook 1 (2), 2465:A / Sax 6, 2761 / Sigma FT-IR 1 (1), 613:B / Structure Index 1, 390:C:1 |
||||||||
| MSDS: | |||||||||
| Description: |
Inhibitor of xanthine oxidase and de novo pyrimidine biosynthesis. A classical agent in treatment of hyperuricemia and gout. |
